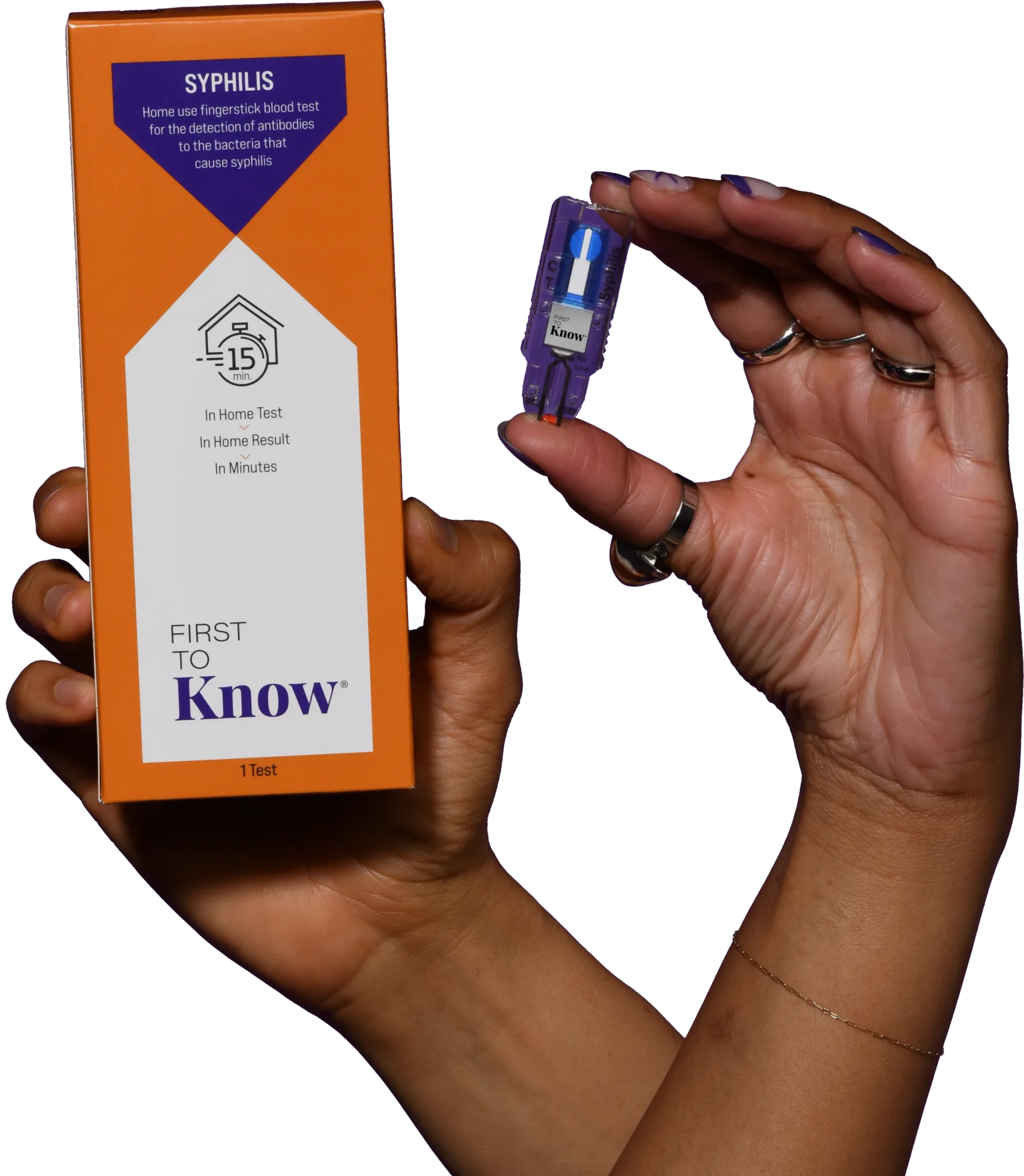
Empowerment through knowledge
What you
don’t know
can hurt you.
Because syphilis symptoms can be hard to notice, detection is critical to protect your health and those you care about.
Buy The TestThe First To Know Syphilis Test
Fast
Get results in as little as 15 minutes.
Accurate
Be confident in your result with clinically proven, patented technology.
Simple
Just touch, tap, test for an accurate result in minutes.
Touch. Tap. Test.
It’s that easy.

Touch
Stick your finger with the provided lancet and touch the Test to your finger to let 1-3 drops of blood enter the Test.
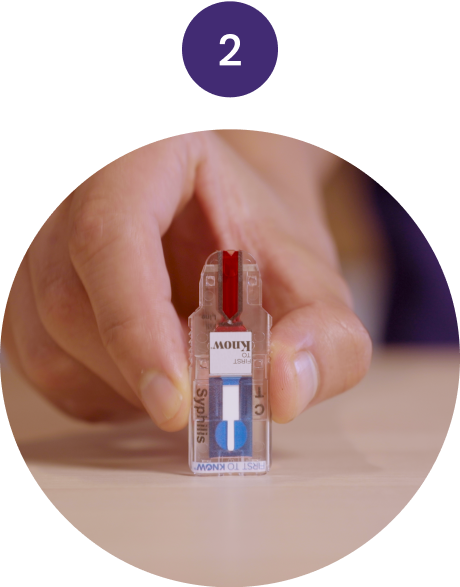
Tap
Tap the Test twice on
a table and lay it flat.
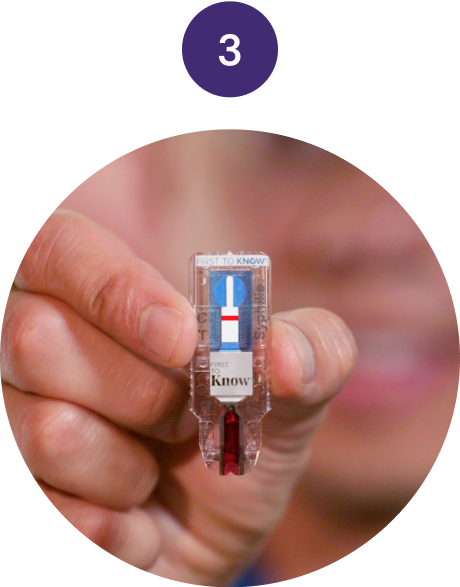
Test
Wait 15 minutes for your
in-home syphilis test result.

Find out now
Your baby could be at risk.
Untreated syphilis during pregnancy can lead to serious health issues for both mothers and babies. Get answers fast to ensure the best start for you and your baby.
Buy The TestFind Out Now
Syphilis has no age limit.
Syphilis cases are increasing across all ages, including older adults. Don’t let it slow you down in your golden years. Take the simple test to get private answers fast.

What you don’t know can hurt you.
Syphilis has been resurging at an alarming rate.
Approx
Increase in syphilis cases in the U.S. over the last 5 years*

What is syphilis?
Syphilis Resources
Syphilis is a sexually transmitted infection (STI) that can cause serious health problems without treatment. It can start with small sores around private parts or in the mouth which may not even be noticed.*
*U.S. Centers for Disease Control and Prevention
Learn MoreIn the news
-
NOWDx to attend the 2025 Minnesota Statewide Conference on HIV June 10-13 in Minneapolis
Read MoreNOWDiagnostics (NOWDx), the manufacturer of the First To Know® Syphilis Test, will be attending the 2025 Minnesota Statewide Conference on HIV June 10-13 in Minneapolis. Visit our booth to see the First To Know® Syphilis Test firsthand – the over-the-counter in-home syphilis test that delivers an accurate result in just 15 minutes. For more conference…
-
NOWDx to attend STI Engage 2025 June 3-5 in Phoenix, Arizona
Read MoreNOWDiagnostics (NOWDx), the manufacturer of the First To Know® Syphilis Test, will be attending STI Engage 2025 June 3-5 in Phoenix, Arizona. Visit Booth #209 to see the First To Know® Syphilis Test firsthand – the over-the-counter in-home syphilis test that delivers an accurate result in just 15 minutes. For more conference details, visit the…
-
NOWDx to attend NASTAD’s 2025 Annual Meeting May 18-21 in Washington, DC
Read MoreNOWDiagnostics (NOWDx), the manufacturer of the First To Know® Syphilis Test, will be attending NASTAD’s 2025 Annual Meeting May-18-21 in Washington, DC. Visit our booth to see the First To Know® Syphilis Test firsthand – the over-the-counter in-home syphilis test that delivers an accurate result in just 15 minutes. For more conference details, visit NASTAD’s…
-
NOWDx to attend 11th Annual NYS Sexual Health Conference May 2 in Canandaigua, NY
Read MoreNOWDiagnostics (NOWDx), the manufacturer of the First To Know® Syphilis Test, will be attending the 11th Annual NYS Sexual Health Conference May 2 in Canandaigua, New York. Visit Booth #6 to see the First To Know® Syphilis Test firsthand – the over-the-counter in-home syphilis test that delivers an accurate result in just 15 minutes. For…
-
NOWDx to attend 2025 NaLa Conference April 29-May 1 in Las Vegas
Read MoreNOWDiagnostics (NOWDx), the manufacturer of the First To Know® Syphilis Test, will be attending the 2025 NaLa Conference in Las Vegas, Nevada April 29-May 1, 2025. Visit Booth #406 to see the First To Know® Syphilis Test firsthand – the over-the-counter in-home syphilis test that delivers an accurate result in just 15 minutes. For more…
-
NOWDx to attend NMAC’s 2025 Biomedical HIV Prevention Summit April 10-12
Read MoreNOWDiagnostics (NOWDx), the manufacturer of the First To Know® Syphilis Test, will be attending the 2025 Biomedical Prevention Summit in Atlanta, Georgia April 10-12, 2025 at Booth #107. Join us to see the First To Know® Syphilis Test firsthand – the over-the-counter in-home syphilis test that delivers an accurate result in just 15 minutes. For…
-
NOWDx to attend AMSUS 2025 Annual Meeting March 3-5
Read MoreNOWDiagnostics (NOWDx), the manufacturer of the First To Know® Syphilis Test, will be attending the AMSUS 2025 Annual Meeting in National Harbor, MD March 3-5, 2025 at Booth #117. Join us to see the First To Know® Syphilis Test firsthand – the over-the-counter in-home syphilis test that delivers an accurate result in just 15 minutes.…
-
First To Know – the only Over-the-Counter Home Test for Syphilis – Will Now Be Available Through the National Alliance of State and Territorial AIDS Directors, Building Healthy Online Communities and the TakeMeHome Initiative
Read More– Program Provides At-Risk Consumers Access to Free Tests Delivered At Home – SPRINGDALE, ARK. – January 22, 2025 – NOWDiagnostics, Inc. (NOWDx), a leader in developing over-the-counter (OTC) and point-of-care (POC) diagnostic tests, proudly announces that its First To Know® Syphilis Test has joined the innovative TakeMeHome initiative. In collaboration with NASTAD (National Alliance…
-
First and Only In-Home Test for Syphilis Now Available to Consumers through Major Retailers and Public Health: NOWDiagnostics Launches First To Know Syphilis Test Across the U.S.
Read MoreWith syphilis cases on the rise, NOWDx’s over-the-counter test offers rapid, in-home results in minutes and is now available nationwide in thousands of pharmacies, retail, and drugstore locations and on Amazon SPRINGDALE, Ark. – November 19, 2024 – NOWDiagnostics, Inc. (NOWDx), a developer of over-the-counter (OTC) and point-of-care (POC) diagnostic tests, today announced that its First…
-
NOWDx to attend the 2025 Minnesota Statewide Conference on HIV June 10-13 in Minneapolis
Read MoreNOWDiagnostics (NOWDx), the manufacturer of the First To Know® Syphilis Test, will be attending the 2025 Minnesota Statewide Conference on HIV June 10-13 in Minneapolis. Visit our booth to see the First To Know® Syphilis Test firsthand – the over-the-counter in-home syphilis test that delivers an accurate result in just 15 minutes. For more conference…
-
NOWDx to attend STI Engage 2025 June 3-5 in Phoenix, Arizona
Read MoreNOWDiagnostics (NOWDx), the manufacturer of the First To Know® Syphilis Test, will be attending STI Engage 2025 June 3-5 in Phoenix, Arizona. Visit Booth #209 to see the First To Know® Syphilis Test firsthand – the over-the-counter in-home syphilis test that delivers an accurate result in just 15 minutes. For more conference details, visit the…
-
NOWDx to attend NASTAD’s 2025 Annual Meeting May 18-21 in Washington, DC
Read MoreNOWDiagnostics (NOWDx), the manufacturer of the First To Know® Syphilis Test, will be attending NASTAD’s 2025 Annual Meeting May-18-21 in Washington, DC. Visit our booth to see the First To Know® Syphilis Test firsthand – the over-the-counter in-home syphilis test that delivers an accurate result in just 15 minutes. For more conference details, visit NASTAD’s…
-
NOWDx to attend 11th Annual NYS Sexual Health Conference May 2 in Canandaigua, NY
Read MoreNOWDiagnostics (NOWDx), the manufacturer of the First To Know® Syphilis Test, will be attending the 11th Annual NYS Sexual Health Conference May 2 in Canandaigua, New York. Visit Booth #6 to see the First To Know® Syphilis Test firsthand – the over-the-counter in-home syphilis test that delivers an accurate result in just 15 minutes. For…
-
NOWDx to attend 2025 NaLa Conference April 29-May 1 in Las Vegas
Read MoreNOWDiagnostics (NOWDx), the manufacturer of the First To Know® Syphilis Test, will be attending the 2025 NaLa Conference in Las Vegas, Nevada April 29-May 1, 2025. Visit Booth #406 to see the First To Know® Syphilis Test firsthand – the over-the-counter in-home syphilis test that delivers an accurate result in just 15 minutes. For more…
-
NOWDx to attend NMAC’s 2025 Biomedical HIV Prevention Summit April 10-12
Read MoreNOWDiagnostics (NOWDx), the manufacturer of the First To Know® Syphilis Test, will be attending the 2025 Biomedical Prevention Summit in Atlanta, Georgia April 10-12, 2025 at Booth #107. Join us to see the First To Know® Syphilis Test firsthand – the over-the-counter in-home syphilis test that delivers an accurate result in just 15 minutes. For…
-
NOWDx to attend AMSUS 2025 Annual Meeting March 3-5
Read MoreNOWDiagnostics (NOWDx), the manufacturer of the First To Know® Syphilis Test, will be attending the AMSUS 2025 Annual Meeting in National Harbor, MD March 3-5, 2025 at Booth #117. Join us to see the First To Know® Syphilis Test firsthand – the over-the-counter in-home syphilis test that delivers an accurate result in just 15 minutes.…
-
First To Know – the only Over-the-Counter Home Test for Syphilis – Will Now Be Available Through the National Alliance of State and Territorial AIDS Directors, Building Healthy Online Communities and the TakeMeHome Initiative
Read More– Program Provides At-Risk Consumers Access to Free Tests Delivered At Home – SPRINGDALE, ARK. – January 22, 2025 – NOWDiagnostics, Inc. (NOWDx), a leader in developing over-the-counter (OTC) and point-of-care (POC) diagnostic tests, proudly announces that its First To Know® Syphilis Test has joined the innovative TakeMeHome initiative. In collaboration with NASTAD (National Alliance…
-
NOWDiagnostics to Present at the 43rd Annual J.P. Morgan Healthcare Conference
Read MoreCEO Robert Weigle to share NOWDx’s vision for transforming consumer healthcare through in-home testing with results in minutes, and highlight commitment to advancing OTC and POC diagnostic products, including novel FDA-authorized First To Know Syphilis Test.
-
First and Only In-Home Test for Syphilis Now Available to Consumers through Major Retailers and Public Health: NOWDiagnostics Launches First To Know Syphilis Test Across the U.S.
Read MoreWith syphilis cases on the rise, NOWDx’s over-the-counter test offers rapid, in-home results in minutes and is now available nationwide in thousands of pharmacies, retail, and drugstore locations and on Amazon SPRINGDALE, Ark. – November 19, 2024 – NOWDiagnostics, Inc. (NOWDx), a developer of over-the-counter (OTC) and point-of-care (POC) diagnostic tests, today announced that its First…
-
NOWDiagnostics and Labcorp Collaborate to Expand Access to the Only FDA-Marketing Authorized Over-the-Counter Syphilis Test in the U.S.
Read MoreAs syphilis cases surge nationwide, the collaboration will bring the First To Know over-the-counter test to clinicians and patients across professional and hospital settings NOWDiagnostics, Inc. (NOWDx), a developer of over-the-counter (OTC) and point-of-care (POC) diagnostic tests, today announced that Labcorp, a global leader of innovative and comprehensive laboratory services, will be the exclusive U.S. distributor…
-
NOWDiagnostics First To Know Syphilis Test Receives FDA De Novo Marketing Authorization for Over-the-Counter Use
Read MoreFirst and only rapid syphilis test with in-home results in minutes, addressing growing epidemic SPRINGDALE, ARK. – August 16, 2024 – NOWDiagnostics, Inc. (NOWDx), a developer of over-the-counter (OTC) and point-of-care (POC) diagnostic tests, today announced that the U.S. Food and Drug Administration (FDA) has granted marketing authorization to its First To Know® Syphilis Test for…